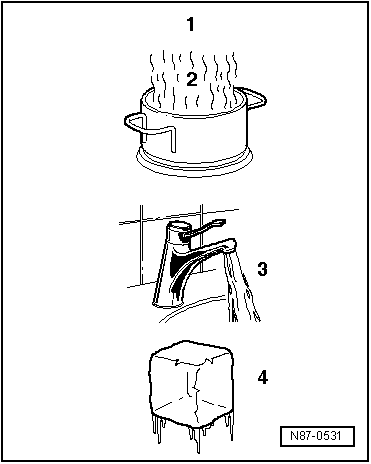
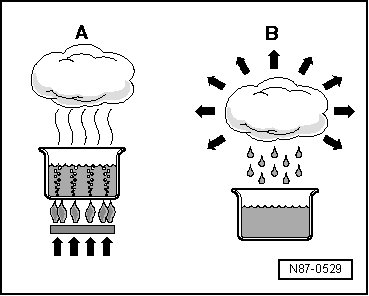
| When water is heated in a vessel (heat absorption), water vapour can be seen to rise. If the vapour is further heated through heat absorption, the visible vapour turns into invisible gas. The process is reversible. If heat is extracted from gaseous water -A-, it changes first to vapour -B-, then to water and finally to ice. |
| Heat always flows from a warmer to a colder substance |
| Every substance consists of a mass of moving molecules. The fast-moving molecules of a warmer substance give off some of their energy to the cooler and thus slower molecules. As a result, the molecular motion of the warmer substance slows down and that of the colder substance is accelerated. This process continues until the molecules of both substances are moving at the same speed. They are then at the same temperature and no further heat exchange takes place. |
|
|
|