| Pressure and boiling point |
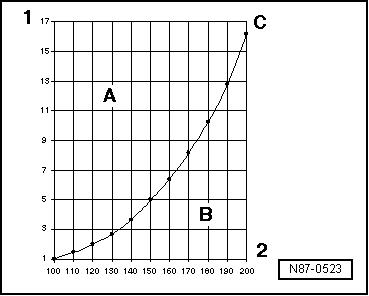
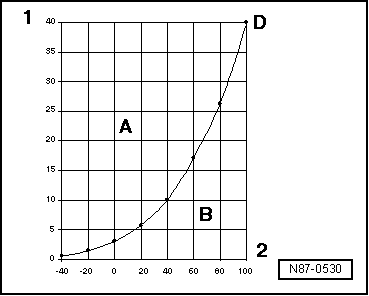
| The boiling point of any liquid in the table of boiling points is always determined for an atmospheric pressure of 1 (one) bar. If the pressure applied on a liquid changes, its boiling point also changes. |
| We know, for example, that at lower pressures water boils at a proportionally lower temperature. |
| By analyzing the characteristic pressure curves of water vapor and R134a refrigerant gas, we find, for example, that vapor turns to liquid at constant pressure when the temperature decreases (in the condenser), and refrigerant gas in liquid state turns to gas when the pressure decreases (evaporator). |
|
|
|