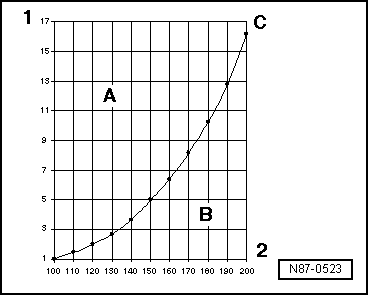
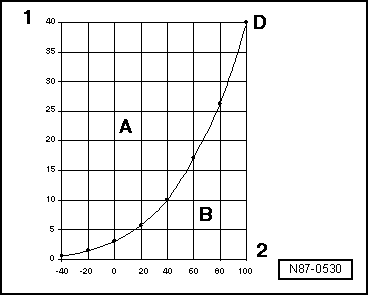
| Pressure and boiling point |
| The boiling point of a liquid given in the tables always refers to the atmospheric pressure of 1 bar. If the pressure of a liquid changes, so does its boiling point. |
| It is known, for example, that water boils at lower temperatures the lower the pressure. |
| Based on the vapour pressure curves for water and refrigerant R134a it can be recognised, for example, that while maintaining pressure, by lowering the temperature, the vapour turns to liquid (in the condenser), or that, for example, by reducing the pressure, the refrigerant turns from the liquid state into the vapour state (evaporator). |
|
|
|